Synthesis and Isolation of a Kinetically Stabilized Crystalline Triangulene | Journal of the American Chemical Society

Homogeneous Metal Salt Solutions for Biomass Upgrading and Other Select Organic Reactions | ACS Catalysis
A' is a blue coloured crystalline salt. On heating it loses blue colour and to give 'B' - Sarthaks eConnect | Largest Online Education Community

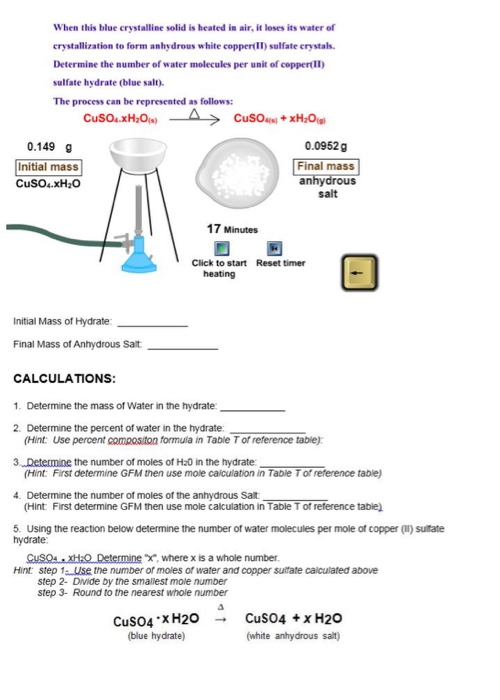
A blue - coloured salt becomes white on heating. Give reason for the above observation. What happens when we add water to the salt obtained after heating ? Also write its formula.

State of the Art in Crystallization‐Induced Diastereomer Transformations - Kolarovič - 2021 - Advanced Synthesis & Catalysis - Wiley Online Library
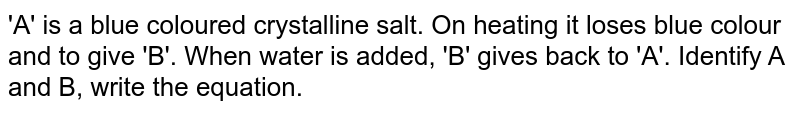
A blue - coloured salt becomes white on heating. Give reason for the above observation. What happens when we add water to the salt obtained after heating ? Also write its formula.

Structural Evolution of Amorphous Precursors toward Crystalline Zeolites Visualized by an in Situ X-ray Pair Distribution Function Approach | The Journal of Physical Chemistry C

Krystol T1 5 gal. Surface-Applied Crystalline Waterproofing Application Krystol T1 K-210 - The Home Depot

A blue - coloured salt becomes white on heating. Give reason for the above observation. What happens when we add water to the salt obtained after heating ? Also write its formula.